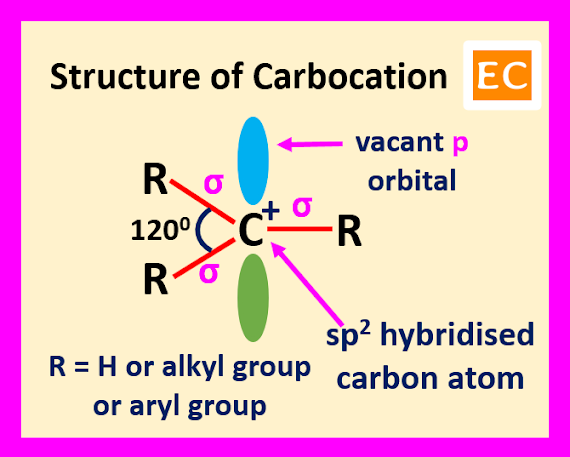
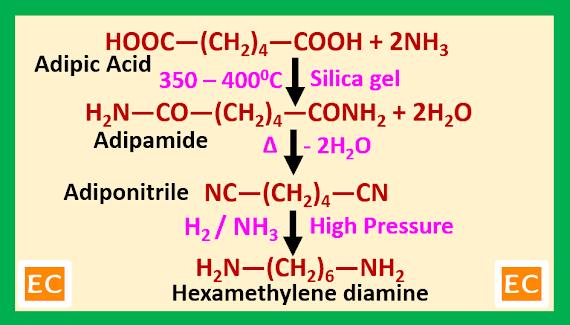
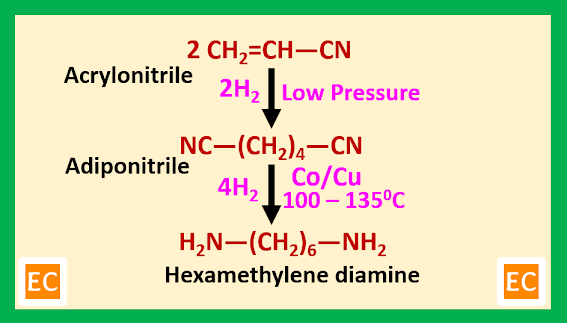
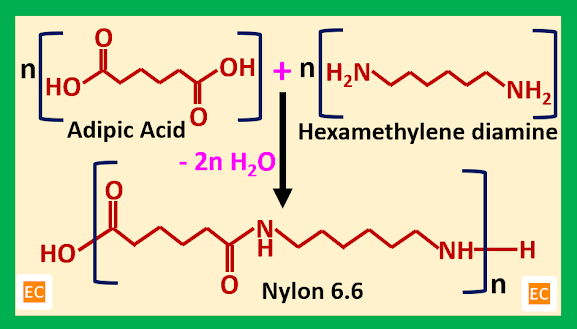
When an
organic species containing a carbon atom and that carbon atom bearing only
three electron pairs, means six electrons is called a carbocation. Carbocation
containing a positive charge and the carbon atom of the carbocation is sp2 hybridized. The carbon atom of the carbocation uses its three hybrid orbitals
for forming three sigma bonds with three substituents and the remaining p
orbital remains vacant. The structure of carbocation is flat structure, having
all the three covalent bonds in one plane. The bond angles between these
covalent bonds are 1200.
Formation of
Carbocation
Carbocation
can be formed in a number of ways. Some of these reactions by which
carbocations are generated are summarized below---
(1) Carbocation is formed by the solvolysis
of C―X bond (where X = halogen such as F, Cl, Br, I or OBs
etc) ---
R―X
-----> R+ + X-
(2)
When alkyl halide (R―X)
reacts with superacids such as SbF5, HSO3F etc,
carbocation is formed --
R―F
+ SbF5 -----> R+ + SbF6-
(3)
When acetyl chloride (R―CO―Cl)
reacts with aluminium chloride (AlCl3) carbocation is generated---
R―CO―Cl
+ AlCl3 ----> R―C+=O + AlCl4-
(4)
When nitrous acid (HNO2)
reacts with amine (R―NH2), carbocation is produced--
R―NH2
+ HNO2 ------> R+ + N2 + H2O +
OH-
(5)
Protonation of alcohol (R―OH)
by acid, followed by dehydration produced carbocation-
R―OH
+ H+ -----> R―OH2+
R―OH2+
-----> R+ + H2O
(6)
When carbonyl compound reacts with H+,
carbocation is formed----
R―CO―R
+ H+ -----> R2C+―OH
(7)
When alkene reacts with H+,
carbocation is formed----
R―CH=CH2
+ H+ -----> R―CH+―CH3
(8)
When allyl halide reacts with Ag+,
allyl carbocation is formed----
CH2=CH-CH2-Br
+ Ag+ ---> CH2=CH-CH2+
+ AgBr
(9)
When benzyl halide reacts with Ag+,
benzyl carbocation is formed-----
Ph―CH2―Br
+ Ag+ ----> Ph―CH2+ + AgBr
Classification
of Carbocation
Carbocations
are classified into three categories----
Primary
Carbocation
When
a positively charged carbon ion linked with another carbon atom, then this
carbocation is called primary carbocation.
CH3―CH2+
Secondary
Carbocation
When
a positively charged carbon ion linked with other two carbon atoms, then this
carbocation is called secondary carbocation.
CH3―CH+―CH3
Tertiary
Carbocation
When
a positively charged carbon ion linked with other three carbon atoms, then this
carbocation is called tertiary carbocation.
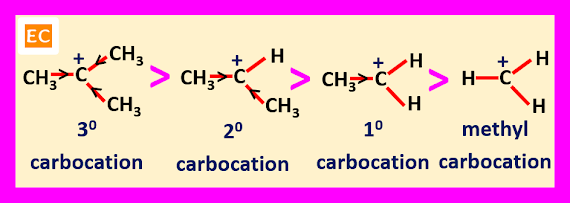
Stability
of Carbocation
The
relative stabilities of the alkyl substituted carbocations are----
R3C+
> R2CH+ > RCH2+ > CH3+
The
stabilities of the alkyl substituted carbocations can be explained by various
factors, which are---
Inductive
Effect
The
alkyl groups have +I effect, so they tend to release electrons and partially
compensate for the electron deficiency of the positively charged carbon. When the number of alkyl groups attached to the positively charged carbon atom
increases, electrons release by this alkyl groups increases and partially compensate for the electron deficiency of the positively charged carbon also
increases. So, the stability of tertiary carbocation (containing three alkyl groups) is greater than secondary carbocation (containing two alkyl groups).
The stability of secondary carbocation is greater than primary carbocation
(containing only one alkyl group). The stability of primary carbocation is
greater than methyl carbocation (containing no alkyl group).
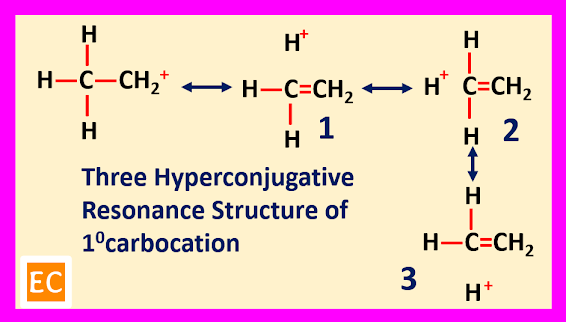
Hyperconjugation
The
sigma (σ) electrons of an alpha (α)
C―H
bond can be delocalized into the unfilled ‘p’ orbital of the positively charged
carbon atom. Thus, spreading the charge over all such bonds. So, several hyper conjugative resonance structure can be drawn for alkyl substituted carbocation and each of
these resonance structure containing same number of covalent bonds as the first
structure.
For
primary carbocation three structure are drawn, for secondary carbocation six
structure are drawn and for tertiary carbocation nine structure are drawn. With increase the number of hyper conjugative resonance structure, the stabilities
of the carbocation increases.
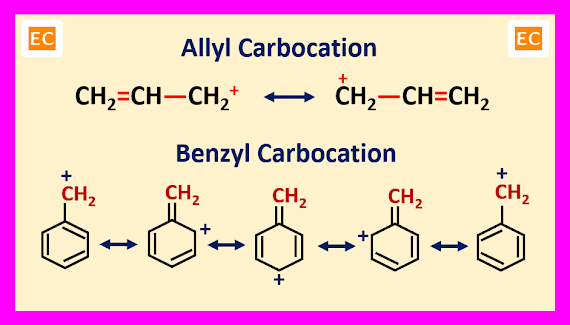
Mesomeric
Effect or Resonance Effect
The
stabilities of conjugated carbocations can be explained by mesomeric effect or
resonance effect. Resonance enhances the stability of a carbocation by delocalization of its charge in conjugated system like allyl or benzyl
carbocation. More number of resonating structures of a carbocation, more will
be its stability. The order of stability of allyl and benzyl carbocations
are----
CH2=CH-CH2+<Ph-CH2+<(Ph)2CH+<(Ph)3C+
Number of resonating structures of allyl carbocation is 2, whereas number
of resonating structures of Ph―CH2+ is 5, (Ph)2CH+ is 9 and
(Ph)3C+ is 13.
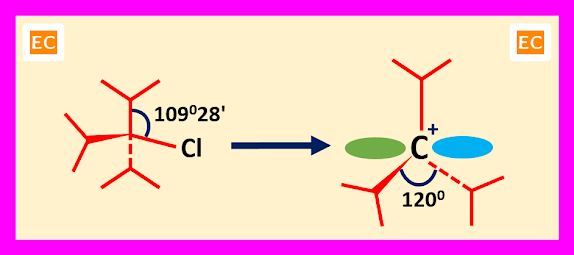
Steric Effect
When carbocations derived from
highly substituted substrate, steric effects play an important role in the
formation and stability of carbocations. Here steric relief is the key factor
for the formation and stability of carbocations. Tri-isopropyl chloride is a highly substituted substrate. In tri-isopropyl chloride three bulky isopropyl
groups are pushed together due to sp3 angle of 109028'. So due to this pushing together, there arises a
strain called B-strain. When the substrate tri-isopropyl chloride ionizes, the
angle expands from 109028' to 1200. So, a relief in
strain arises due to this angle expands and space between alkyl groups
(isopropyl groups) increases.