Raman Spectroscopy
Quantum Theory of Raman Scattering
Classical Theory of Raman Scattering
E = E0 sin 2πνt ----- (4)
Where E0 is the amplitude of the vibrating electric field vector and v is the frequency of of the incident light radiation. Thus from equation (3) and (4), we get
µ = αE0 sin 2πνt ----- (5)
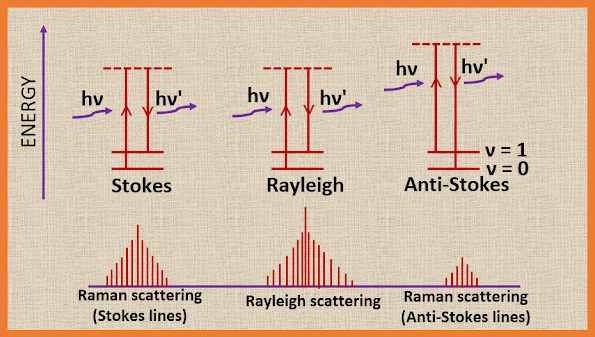
Such an oscillating dipole emits radiation of its own oscillation with a frequency v, giving the Rayleigh scattered beam, if , however, the polarizability varies slightly with molecular vibration, we can write
α = α0 + (Әα/Әq) q ----- (6)
Where the coordinate q describe the molecular vibration. We can also write q as
q = q0 sin 2πνmt ----- (7)
Where q0 is the amplitude of the molecular vibration and vm is its frequency. From equation (6) and (7), we have
α = α0 + (Әα/Әq) q0 sin 2πνmt ----- (8)
Substituting for α in equation (5), we have
µ = α0E0 sin 2πνt + (Әα/Әq) q0E0
sin 2πνt sin 2πνmt ----- (9)
Making use of trigonometric relation :
sin x sin y = 1/2 [cos (x-y) - cos (x+y)]
This equation reduces to
µ = α0E0
sin 2πνt + 1/2 (Әα/Әq) q0E0 cos 2π (ν-νm) t – 1/2
(Әα/Әq) q0E0 cos 2π
(ν +νm) t ----- (10)
= α0E0 sin 2πνt + 1/2 (Әα/Әq) q0E0
x [cos 2π (ν-νm) t – cos 2π (ν+νm) t] ----- (11)
Thus the oscillating dipole has three distinct frequency components : (i) the exciting frequency v with amplitude α0E0 (ii) v-vm and (iii) v+vm with very small amplitudes = (1/2) (Әα/Әq) q0E0. Hence, the Raman spectrum of a vibrating molecule consists of a relatively intense band at the incident frequency and two very weak bands at frequencies slightly above and below that of the intense band.
If, however, the molecular vibration does not change the polarizability of the molecule, then (Әα/Әq) = 0 so that the dipole oscillates only at the frequency of the incident (exciting) radiation. The same is true of the molecular rotation. We conclude that for a molecular vibration or rotation to be active in the Raman spectrum, it must cause a change in the molecular polarizability, i.e., Әα/Әq ≠ 0. --- (12)
Homonuclear diatomic molecules such as H2, N2, O2, which do not show IR spectra because they do not possess a permanent dipole moment, do show Raman spectra since their vibration is accompanied by a change in polarizability of the molecule. As a consequence of the change in polarizability, there occurs a change in the induced dipole moment at the vibrational frequency.
Rotational Raman Spectra
For the vibrational Raman spectrum of a diatomic molecule, the selection rule is
∆ν = ±1 ----- (13)
For the rotational Raman spectrum of a diatomic molecule, the selection rule is
∆J = 0, ±2 ----- (14)
For the rotational selection rule, the operative part is
∆J = +2 ----- (15)
Since ∆J = 0 corresponds to Rayleigh scattering and ∆J = -2 transition can be ignored as the rotational quantum number of the upper state must be greater than that of the lower state. Using the selection rule with the energy level expression for the rigid diatomic rotor, viz,
F(J) = BJ (J+1) cm-1 (J = 0, 1, 2, 3 ---)
We find that ∆F(J) = F(J+2) - F(J) = B (4J+1) cm-1
Since ∆J = +2, we may level these lines S branch lines and thus,
∆F(J) = B(4J+6) cm-1 (J = 0, 1, 2, 3 ---) --- (16)
Where J is the rotational quantum number in the lower state.
Thus if the rotational energy gains by the molecule from the photon during collision we have a series of S branch lines to the low wavenumber side of the exciting line (Stokes line), while if the molecule loses energy to the photon the S branch lines appear on the high wavenumber side (anti-Stokes lines). The wavenumber of the corresponding spectral lines are given by :
ν̅s = ν̅exc ± ∆F (J) = ν̅exc ± B
(4J +6) cm-1 ----- (17)
Where the plus(+) sign refers to anti-Stokes lines, the minus (-) sign to Stokes lines, and ν̅exc is the wavenumber of the exciting radiation.
The energy level diagram and the rotational Raman spectrum for a diatomic molecule are shown in the figure. We see that the first Stokes line (or the first anti-stokes line) appears at a distance of 6B from the exciting Rayleigh line. This is evident from equation 16, where putting J=0 we obtain ∆F (J) = 6B. The separation of the successive Raman lines is, however, 4B, as can be easily verified by putting J = 1, 2, 3, etc. in equation 17.
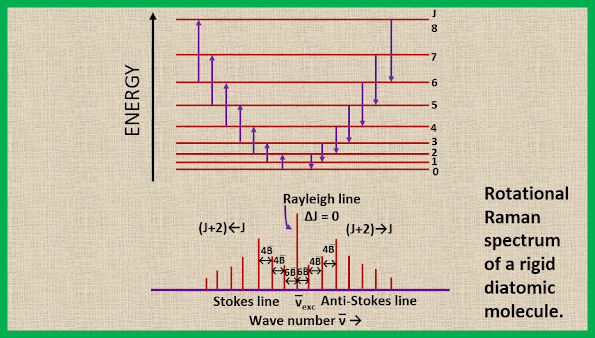 |
| Rotational Raman spectrum of a rigid diatomic molecule |
Rotation-Vibration Raman Spectrum
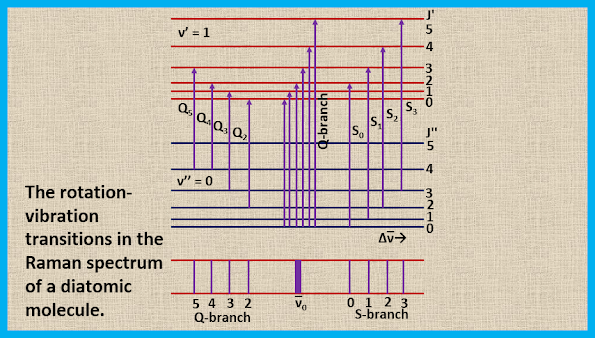
For the rotation-vibration Raman spectrum, the selection rules involve the changes in both the vibrational and the rotational quantum numbers. For the rotation vibration Raman spectrum of a diatomic molecule the selection rules are:
∆ν = +1; ∆J = 0, ±2 ----- (18)
Since at room temperature most of the molecules are in the ground vibrational state (v = 0), so we see, only the vibrational transition from v = 0 to v = 1. The transition with ∆J = 0 form a Q-branch, the transition with ∆J = +2 form an S-branch, and the transition with ∆J = -2 form an O-branch. The rotational Raman transitions accompanying a 0→1 vibrational transition are shown in the figure. Here, ∆ν̅ measures the displacement from the exciting mercury line. The Q-branch exhibits an intense narrow line at ν̅exc ± B that is usually unresolved while the S and O branches form weak wings which extend to lower and higher wave numbers, respectively, from the intense narrow line.
The rotation-vibration transitions in the Raman spectrum of a diatomic molecule











No comments:
Post a Comment