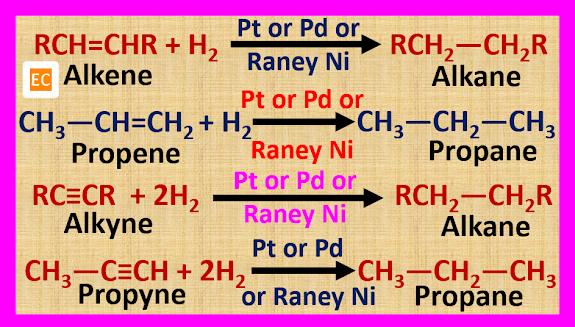
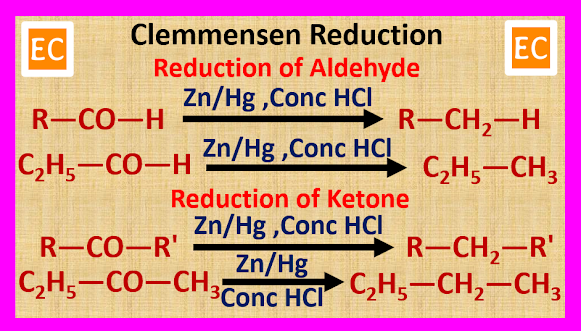
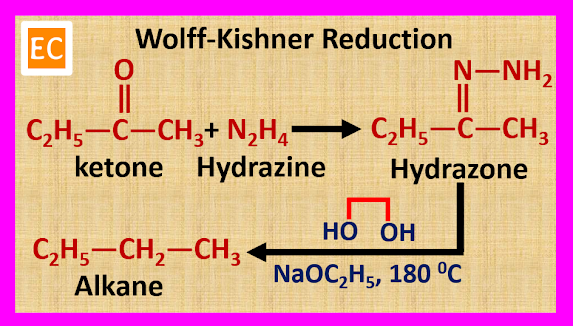
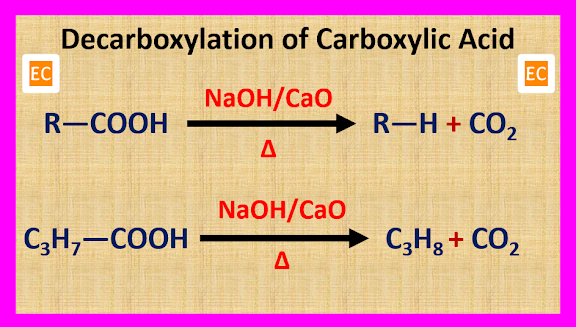
ULTRAMARINE BLUE
Synthetic ultramarine blue is the most widely used blue pigment in the present time. Ultramarine blue pigment is insoluble in water and dark blue in colour. In 1928, blue ultramarine was first prepared by Guiment and Gamelin. The ultramarine blue pigment is a complex silicate of aluminium and sodium assisted with about 12% sulphur.
Blue, green and white are three varieties of ultramarine. Among these three varieties, blue colour variety is the most popular and this blue ultramarine is used as a pigment. This blue colour variety can be prepared from green or white ultramarine. These ultramarine pigments are affected by acid but stable in alkali and light. Due to liberation of H2S gas, these pigments are affected by acid. The composition of three types of ultramarine are as follows---
Blue ----- Na5Al3Si2S3O12
Green ----- Na5Al3Si2S2O12
White ----- Na5Al3Si3SO12
The ultramarine composition is however not stoichiometric. Because the degree of exchange that takes place depends upon the various factors. These factors are solution concentration, heating time etc.
Preparation of Blue Ultramarine
When a mixture of sodium carbonate (soda ash), kaolin, charcoal, quartz, sulfur and resin is heated blue ultramarine is prepared. [It is very important that iron should not present in the raw materials. If iron present in the raw materials it appreciably dulls the colour and forms a white mass. In presence of air this white mass changes to green.] After heating, the product is cooled and powdered. If sodium carbonate (soda ash) in the raw materials is substituted by sodium sulphate, then darker blue colour is produced.
For the manufactured of ultramarine there are three methods--
(1) Sulphate process--- In the sulphate process kaolin, sodium sulphate and charcoal are heated together.
(2) Soda sulphate process--- In the soda sulphate process kaolin, sodium carbonate (soda ash), sodium sulphate, sulfur and tar are heated together.
(3) Soda process--- In the soda process kaolin, sodium carbonate (soda ash), sulfur, colophonium and tar heated together.
The depth of the colour of the ultramarine increases with the increase in the amount of sulfur content. In the ultramarine, sulfur present is in the same form as in the polysulphides, is the probable reason for the colour of ultramarine. In the blue ultramarine Na+ ion is present. When Na+ ion is replaced by other cations such as K+ ion or Ag+ ion, it is found that the colour of ultramarine changes to various colour, depending on the cations. This indicates that within the alumino silicate skeleton, the cation present, have a potential influence on the colour.
Uses of Ultramarine
(1) Ultramarine blue is widely used for wall painting.
(2) It is used for laundry purpose. It is used for neutralize the yellowish tone in cotton or linen fabrics.
(3) For painting woods ultramarine blue used as a pigment.
(4) For whitening, paper and other products, ultramarine is also used.
Limitation
Ultramarine can not be used as a cooling material or iron articles because in ultramarine large percent of sulfur present. Due to possibility for the formation of PbS, it can not be mixed with lead pigments.